NICTN Information for Professionals
This area of the website will expand over time to include more resources for researchers.
For any further information please contact us at the NICTN Co-ordinating Centre at nictn@belfasttrust.hscni.net.
Study Adoption to the NICTN Portfolio
The NICTN undertakes a Review and Adoption process before any clinical trial (or other clinical or translational research study of high quality design) is adopted into the NICTN portfolio. Adoption to the NICTN portfolio is an essential step for access to research support from NICTN staff or services to facilitate trial set-up and delivery.
To initiate the review and adoption process Investigators should complete a NICTN Portfolio Adoption and Registration Application Form, available on request to nictn@belfasttrust.hscni.net.
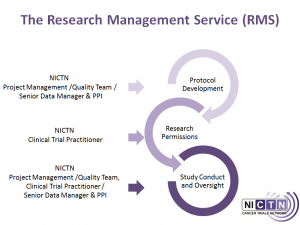
The NICTN Research Management Service (RMS)
The NICTN RMS is a service that is additional to the core services of the NICTN (see section ‘About Us’). The RMS facilitates protocol development, research permissions, study conduct and oversight for small, investigator-led studies from investigators based in Northern Ireland with a local HSC or academic sponsor.
Background to the NICTN RMS
- A service provided by the NICTN to support quality delivery of local clinical research
- Operates within the HSC R&D infrastructure
- Management throughout the study lifecycle
- Independent of site function
- 3 Key Dimensions of Support
- Project Management
- Data Management
- Regulatory Management
Scope of the RMS
- HSCT or local academic sponsor
- Investigator-led
- Single-centre/multi-centre (NI only)
- Studies can be in adults or children, blood cancers or solid tumours and in pre-malignant disease as well as invasive malignancies. However, we may not have adequate expertise for certain methodologies or diseases.
Note: The scope of the RMS excludes studies managed by the Northern Ireland Clinical Trials Unit (NICTU) or any other accredited CTU. NICTN RMS does not include any formal statistical support, and requests for this should be made to the NI Methodology Hub who are providing a statistical and methodological ‘sign-posting and advice service’ for HSC researchers.
Objectives of the RMS
- More guidance and templates for researchers
- More seamless communication
- Efficient use of resources
- Shorter timelines
The RMS in Practice
Potential Wider Impact of the RMS
- High quality research / maximum patient benefit
- High impact publications
- Promotes progressive research culture
- Timely clinical innovation
- Successful inspection outcomes
- Positive reputation and funding impact
Personal and Public Involvement in Cancer Research – The NI Cancer Research Consumer Forum
The NI Cancer Research Consumer Forum, supported by NICTN, is a group of people affected by cancer who have a passion for Personal and Public Involvement (PPI) in cancer research. Members are committed to the goal of PPI which is to maximise patient benefit through high quality research. This is achieved through researchers and patients/carers working in partnership, across the research cycle.
The Forum was established in 2011 and since then it has been involved in PPI activities with a number of researchers and research bodies and institutions. Members have been involved in all or some of the following:
- Research funding co-applicant
- Reviewing and advising on clinical research protocols and patient information leaflets
- Research steering group or committee membership
- Trial Management Group membership
- Advising on the best methods and information to raise patient and public awareness about cancer clinical trials available in Northern Ireland
- Promoting PPI in cancer research
If you are undertaking cancer research and wish to work with the Forum, please contact Ruth Boyd, NICTN PPI Professional Lead, at nictn@belfasttrust.hscni.net.
Investigator GCP and CV requirements
National Institutes of Health Research GCP Training
The website below can be used for creating a new account or accessing an NIHR account to undertake GCP training (secondary care option).
http://learn.nihr.ac.uk/mod/certificate/view.php?id=286
(accessed 04 July 2017)
NICTN CV Template
Please see NICTN CV Template for completion. Other CV formats can also be submitted if they incorporate the template information.